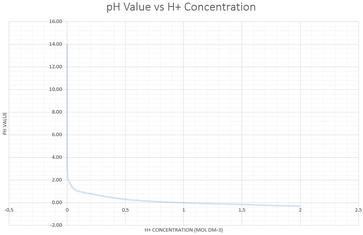
The pH of a solution describes its acidity and is the negative logarithm ( log) of its hydrogen ion concentration. The term pH is used because the hydrogen ion concentration in solutions of weak acids and in many other fluids is frequently much less than 1 ...

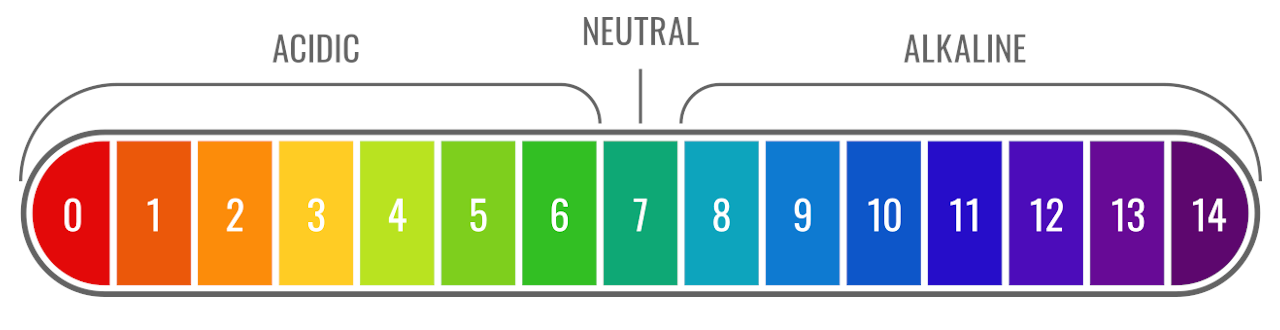
The pH scale The pH scale is used to measure how acidic or basic a solution is. The lower the pH, the more acidic the solution. The higher the pH, the. -
![pH = -log[H+] assuming 100 percent dissociation; if given percent ionization, multiply by the molarit… | Chemistry lessons, Chemistry education, Chemistry classroom pH = -log[H+] assuming 100 percent dissociation; if given percent ionization, multiply by the molarit… | Chemistry lessons, Chemistry education, Chemistry classroom](https://i.pinimg.com/originals/99/80/05/998005d7b3fbb74f7a91222f3209e7c5.png)





![Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC" Finding the pH, pOH, [H+], [OH-] - ACIDS AND BASES: IT'S ACTUALLY Quite "BASIC"](http://itsactuallyquitebasic.weebly.com/uploads/2/7/8/0/27808159/4950515.png?357)
![Solving For pH, pOH, [H+], [OH-] - Acids & Bases Solving For pH, pOH, [H+], [OH-] - Acids & Bases](http://youarebasic.weebly.com/uploads/5/0/1/4/50143245/5802360_orig.png)

![pH = -log[H+] pH = -log[H+]](https://s3.studylib.net/store/data/008427242_1-6a856521ca6b3bcc442fa00c77f4fe6e.png)
![Calculate [H+] from pH - YouTube Calculate [H+] from pH - YouTube](https://i.ytimg.com/vi/yE4M2Uuh-h4/sddefault.jpg)



![Calculations of pH, pOH, [H+] and [OH-] Calculations of pH, pOH, [H+] and [OH-]](https://www.sciencegeek.net/Chemistry/taters/graphics/pHSchematic.gif)


![Given [H+] or [OH-], Calculate pH & pOH - YouTube Given [H+] or [OH-], Calculate pH & pOH - YouTube](https://i.ytimg.com/vi/ghIYaqo0Ycc/maxresdefault.jpg)



