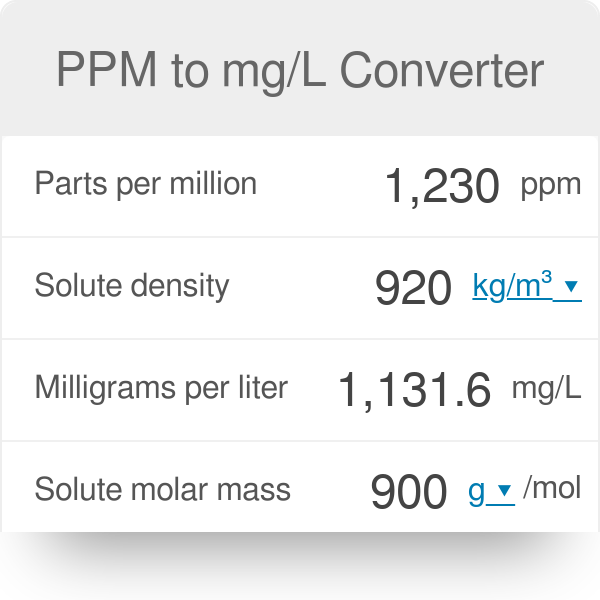
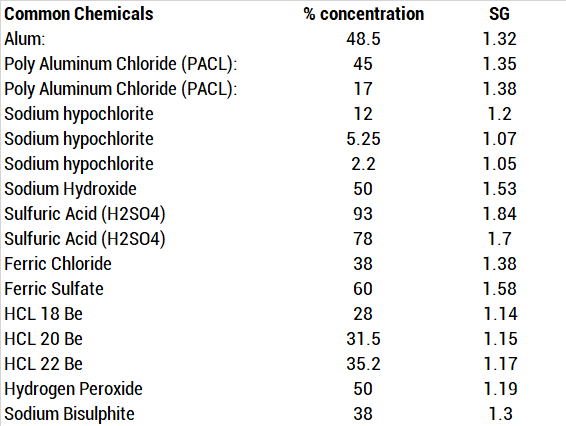
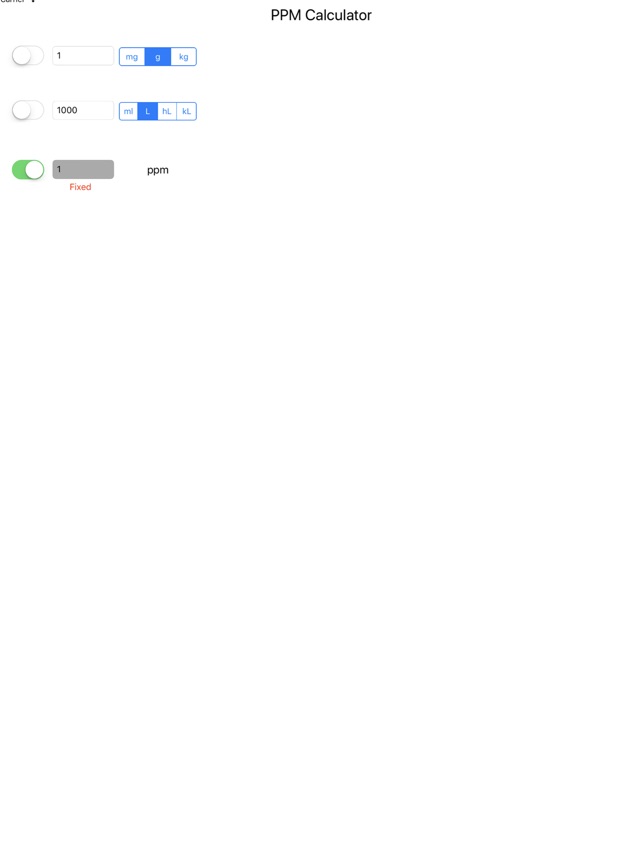
SOLVED: Calculate the concentration in ppm of HCl solution having a density of 1.019 g/mL and contains 0.362% by mass HCI.

Solution of 100 ml water contains 0.73 g of Mg(HCO(3))(2) and 0.81g of Ca(HCO(3))(2) calculate the hardness in terms of ppm of CaCO(3)