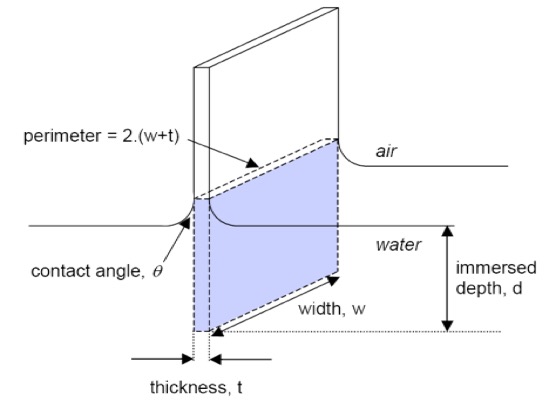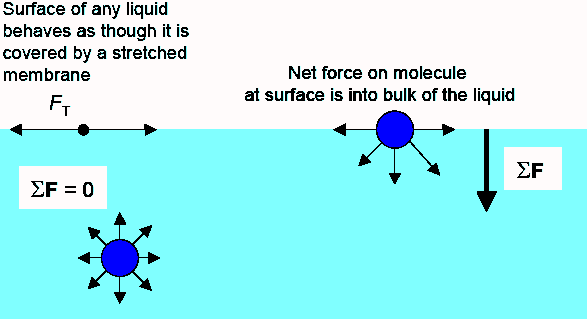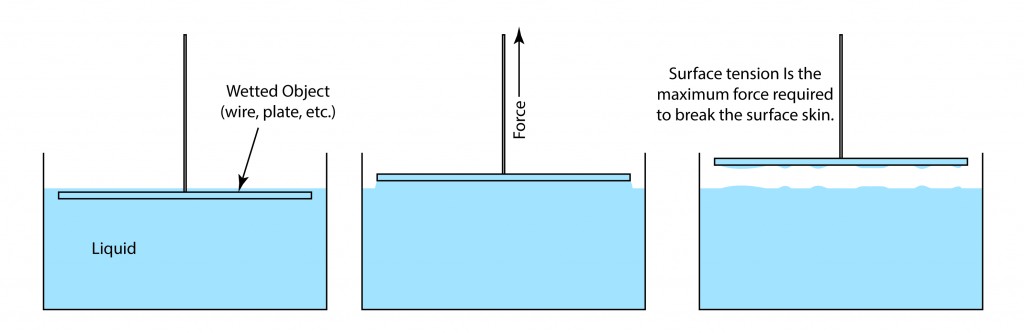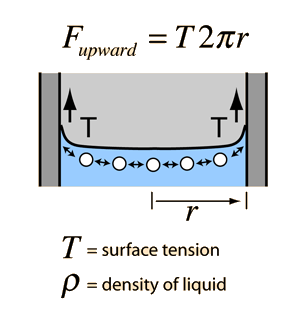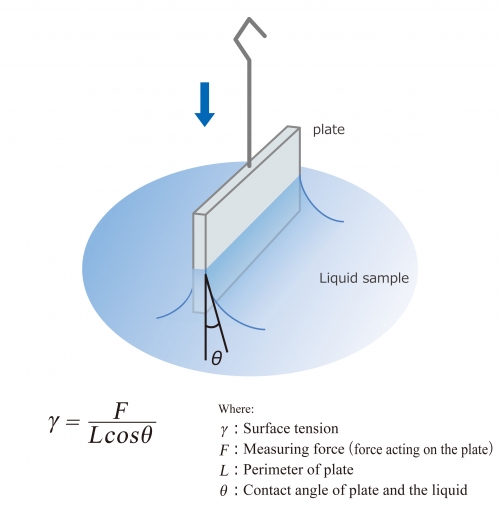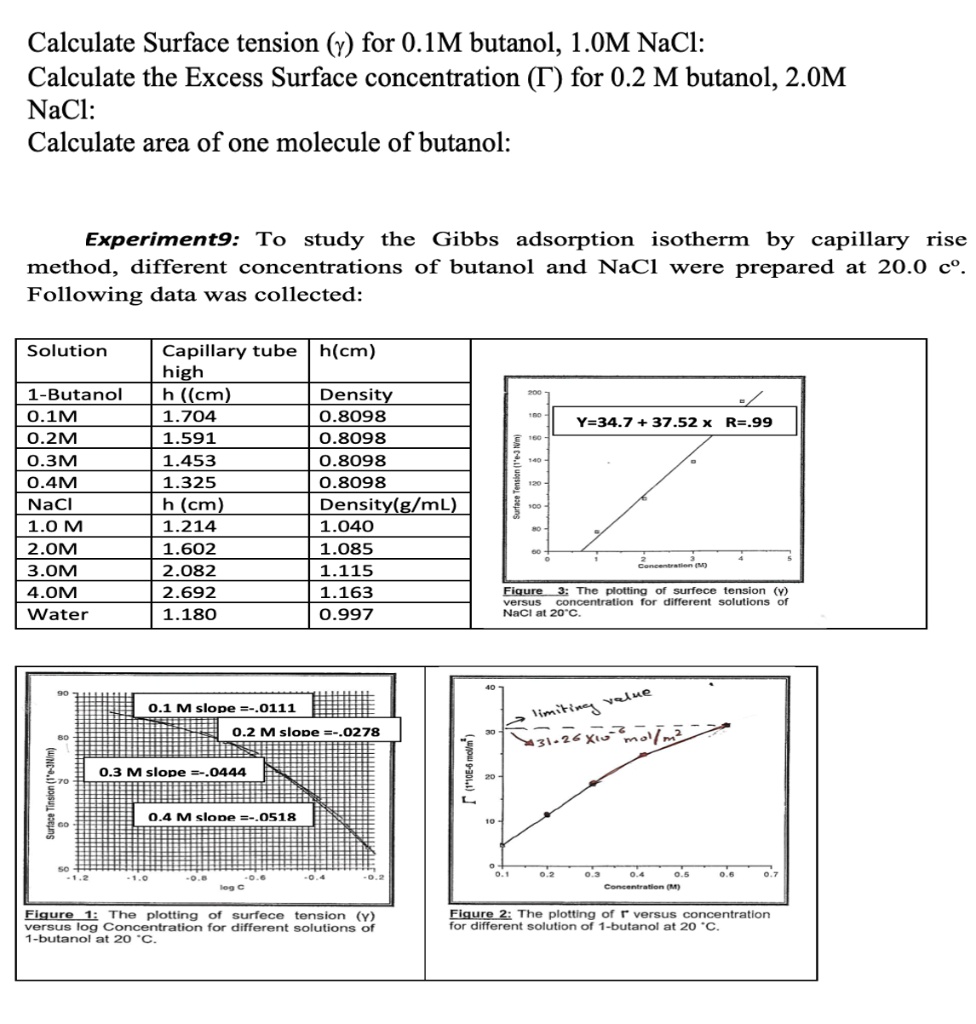
SOLVED: Calculate Surface tension for 0.IM butanol, 1.OM NaCl: Calculate the Excess Surface concentration (T) for 0.2 M butanol, 2.OM NaCl: Calculate area of one molecule of butanol: Experiment9: To study the

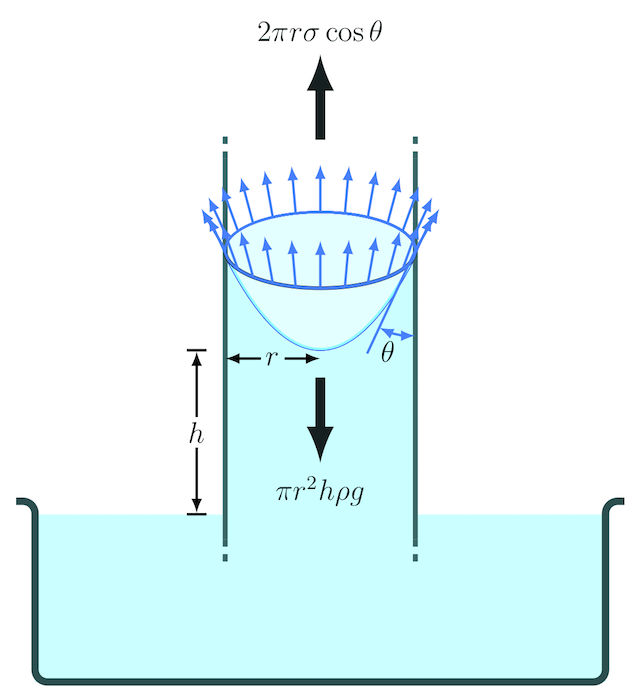
Calculate the rise of water inside a clean glass capillary tube of radius 0.1 mm , when immersed in water of surface tension 7 × 10^-2 N/m . The angle of contact

Surface tension of water is 0.072N/m . The excess pressure inside a water drop of a diameter 1.2 mm is:

The surface tension of water at 20^(@)C is 73 dynes cm^(-1), The minimum value of work done needed to increases surface are of water from 2 cm^(2) to 5cm^(2) is

11.8 Cohesion and Adhesion in Liquids: Surface Tension and Capillary Action – College Physics chapters 1-17

Using data from the diagram below, calculate the ratio of the height to which water at 0 C and mercury are raised or suppressed by capillary action in the same glass tube. (

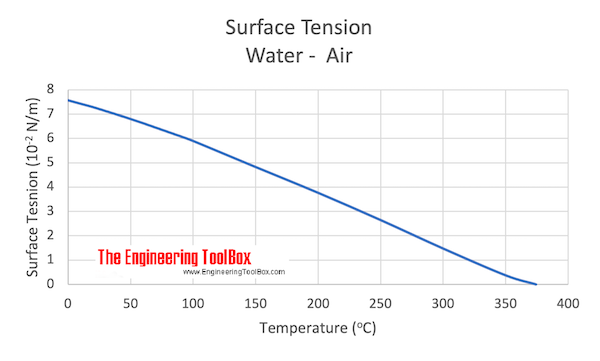
![Values of the surface tension of water at different temperatures [1]. | Download Table Values of the surface tension of water at different temperatures [1]. | Download Table](https://www.researchgate.net/publication/231143777/figure/tbl1/AS:668494844686342@1536392999595/Values-of-the-surface-tension-of-water-at-different-temperatures-1.png)