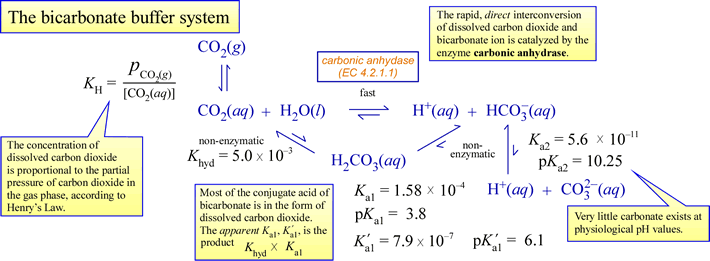
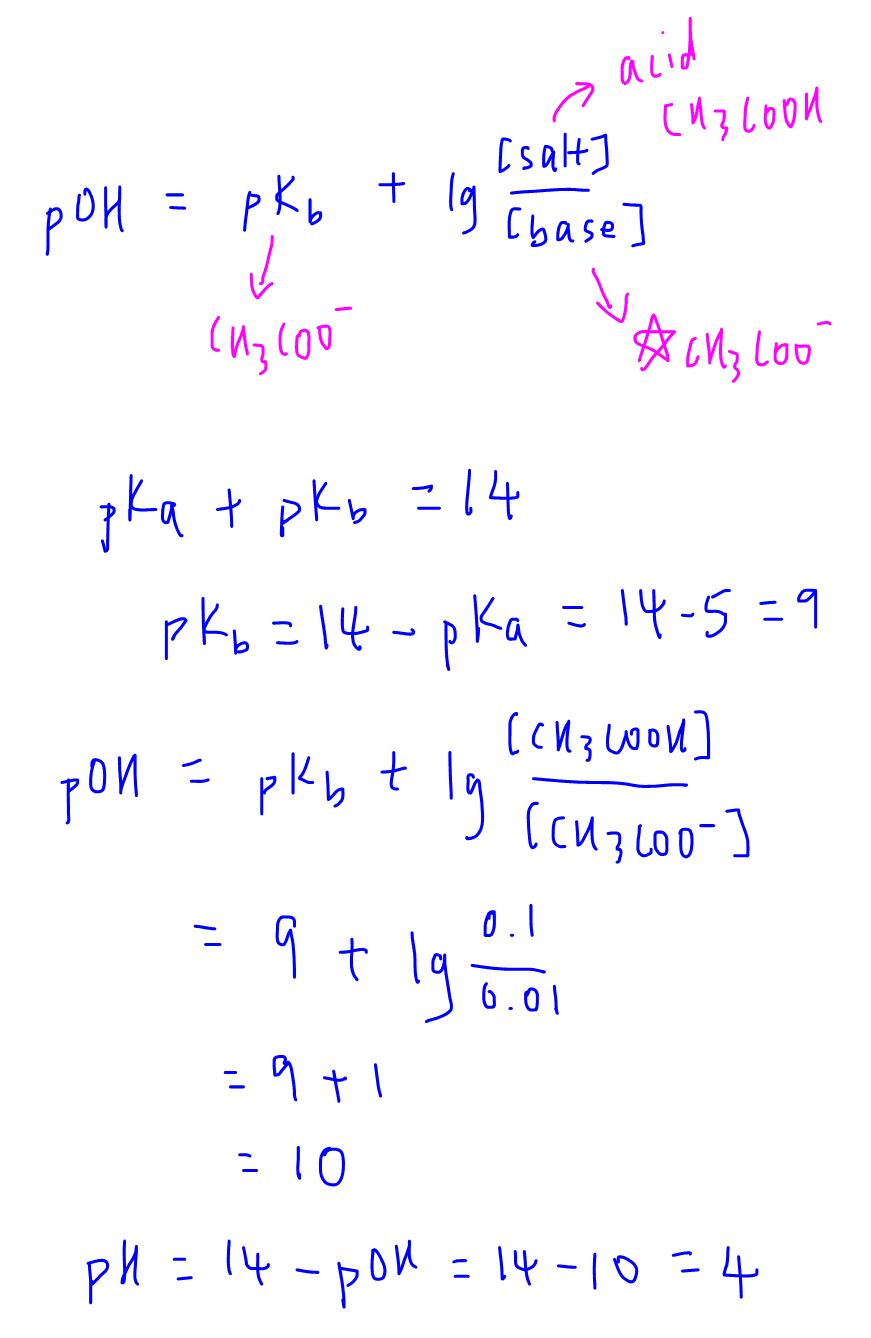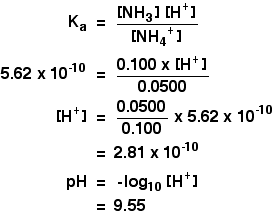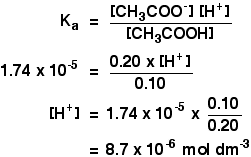Preparation of Buffers - 1 Calculate the volume of sulfuric acid (H 2 SO 4 ) necessary to prepare 600 milliliter 0.5M H 2 SO 4 from concentrated H 2 SO. - ppt download

SOLVED: You will be preparing a working solution of 1X using a stock solution of 100X buffer. Using the formula below, perform the calculation for preparing 250 mLs of a 1X working
![Calculate pH of a buffer prepared by adding 10 mL of 0.10 M acetic acid to 20 mL of 0.1 M sodium acetate. [pKa (CH3COOH) = 4.74 ] Calculate pH of a buffer prepared by adding 10 mL of 0.10 M acetic acid to 20 mL of 0.1 M sodium acetate. [pKa (CH3COOH) = 4.74 ]](https://i.ytimg.com/vi/t9B5VgPOTG4/maxresdefault.jpg)