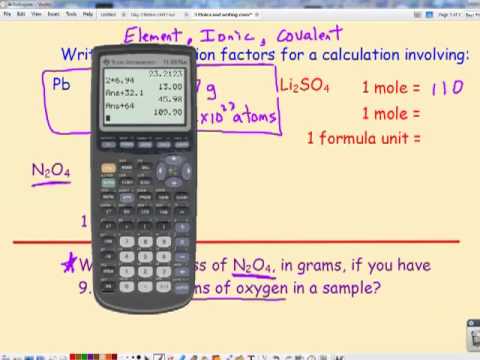
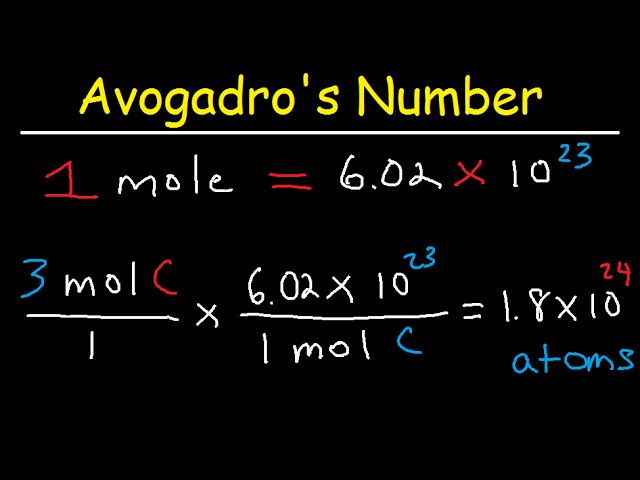Objective: To reintroduce the mole and introduce two mole conversions Do Now: Solve each proportion = x2. 10 = x. - ppt download

Calculate the Avogadro's number using the charge on the electron `1.60 xx 10^(-19)C` and the fact - YouTube

Casio fx-991EX calculator: HOW TO ACCESS CONSTANTS (Avogadro's constant, Planck's constant, etc.) - YouTube

Converting Between Particles and Moles Using Avogadro's Number and Conversion Factors – Science and Joe

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5
Is there a preset function in Texas Instrument-84 Plus calculators for Avogadro's number? Can you add one? - Quora

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5